Nemera’s metering dose pump and actuator for Flixonase Allergy Relief, approved by NMPA (National Medical Products Administration) and launched in China!



As a partner-of-choice, Zollner will support the design, development and manufacturing of electronic drug delivery systems for both, Nemera’s proprietary and customer owned products. This collaboration will begin with Nemera’s...
Continue Reading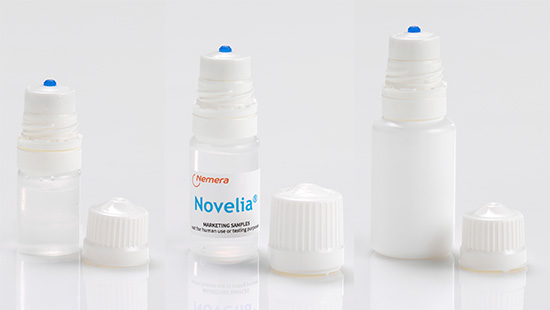
The Center for Drug Evaluation (CDE) performs regulatory evaluation of new drugs and medical devices to assess if these can be brought to the market in China. Our multi-dose eye...
Continue Reading
SciTropin A™ Pen is a state-of-the-art pen injector device manufactured by Nemera. It was included in the ARTG in February 2022 opening the way for the device to be distributed...
Continue Reading
Yesterday, we laid the first stone of our new state-of-the-art manufacturing facility in Szczecin, Poland. This facility will help extend our manufacturing capabilities and bring more drug delivery device solutions...
Continue Reading
Nemera metering dose pump and actuator for Flonase Allergy Relief has been approved by the U.S. FDA Nemera, one of the world leaders in the design, development and manufacturing ...
Continue Reading
Nemera awarded silver medal in the EcoVadis sustainability assessment, figuring in the top 22% of all companies evaluated. At Nemera we are committed to conducting business in an ethical and...
Continue Reading